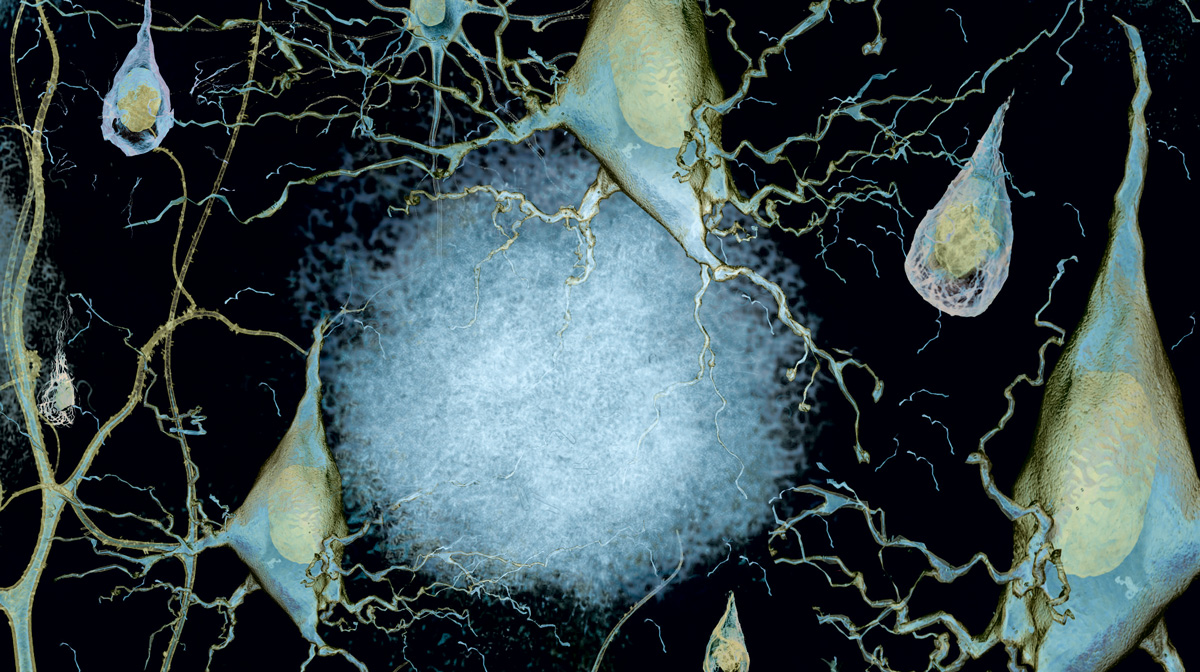
If one could peer down at a healthy brain’s electrical circuitry, it would resemble a tremendous city at night, illuminated by myriad highways and traffic. One would see a grid of three-dimensional complexity, marked by wispy branches of neurons reaching to each other and neurons firing signals across junctions called synapses. It would be a scene of beautiful interdependence.
As Veronica Galvan, Ph.D., assistant professor of physiology at the UT Health Science Center’s Sam and Ann Barshop Institute of Longevity and Aging Studies, puts it: “The neurons live to be connected; an isolated neuron will not live. The neurons talk to each other across long distances.”
Alzheimer’s disease, the memory-robbing disease associated with aging, mars that landscape with unwanted plaques and tangles of proteins. The disease dampens the synaptic signals, bringing the song of the neurons to a close in some quadrants. Over time, the synapses disintegrate from lack of use and affected neurons die. “Once you lose these networks, complete systems fall apart,” Dr. Galvan says. “That is how people first lose short-term memory, then have alterations in personality and, finally, move into frank dementia.”
Dr. Galvan and a fellow assistant professor, Salvatore Oddo, Ph.D., joined the Health Science Center Department of Physiology and Barshop Institute in 2008. Both are making intriguing observations in mice about what takes place in the earliest events of Alzheimer’s, long before cognitive deficits become apparent. The early changes are thought to be the most toxic, the most essential to understand and the most addressable. “We, and I think most in the field, have a strong interest in figuring out the initial stages of the disease, when the connections start to go,” Dr. Galvan says.
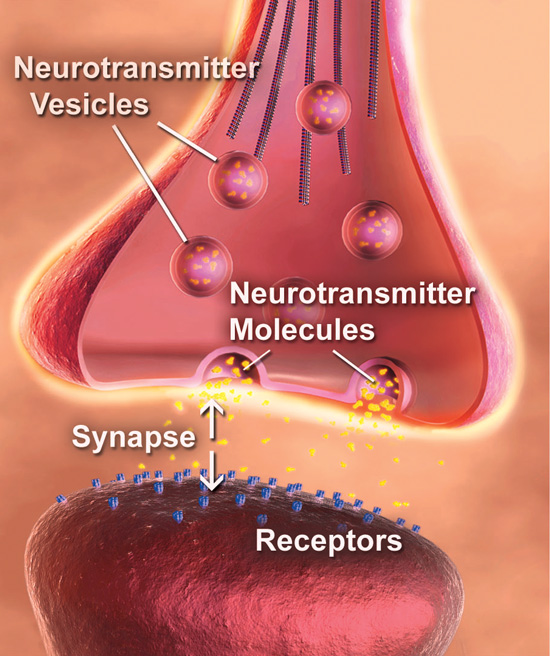
Alarming Alzheimer’s disease statistics
Source: Dr. Salvatore Oddo
- 5 million people in the U.S. have Alzheimer’s disease.
- 40 percent of people age 85 and older are at risk for Alzheimer’s disease.
- Alzheimer’s is the seventh-leading cause of death in the U.S.
- The prevalence of Alzheimer’s disease in the U.S. and Texas is expected to double by 2050.
Dr. Oddo in 2003 published a paper describing a new mouse model of Alzheimer’s disease. Today this model is used in 120 laboratories worldwide because it paints a more comprehensive portrait of Alzheimer’s than the other mouse models. In the “triple-transgenic mouse,” Dr. Oddo and his colleagues inserted three genes that caused the model to develop both lesions seen in the brains of human Alzheimer’s patients. One lesion is called amyloid-beta plaques and the other is called tau-protein tangles. All previous mouse models of Alzheimer’s developed only the amyloid-beta plaques, Dr. Oddo says. He is studying the relationship of the two types of lesions and has found that the amyloid-beta accumulation leads to the tau tangles, whereas tau does not result in the amyloid deposits. “We are trying to find out what it is about the accumulation of these two proteins that leads to learning and memory deficits,” he says.
Dr. Galvan studies an Alzheimer’s mouse model that has the same rare genetic mutations found in human families in Sweden and Indiana. In these families, half of the brothers and sisters develop Alzheimer’s with very early onset. This inherited form of the disease represents only 1.5 percent of Alzheimer’s cases but provides clues to the processes that cause the disease. “The mouse model that we use mimics this familial Alzheimer’s situation,” Dr. Galvan says. “So we have little mouse families in which there are brothers and sisters, and some of them develop the disease and some won’t, and it segregates 50-50 exactly like the human families.”
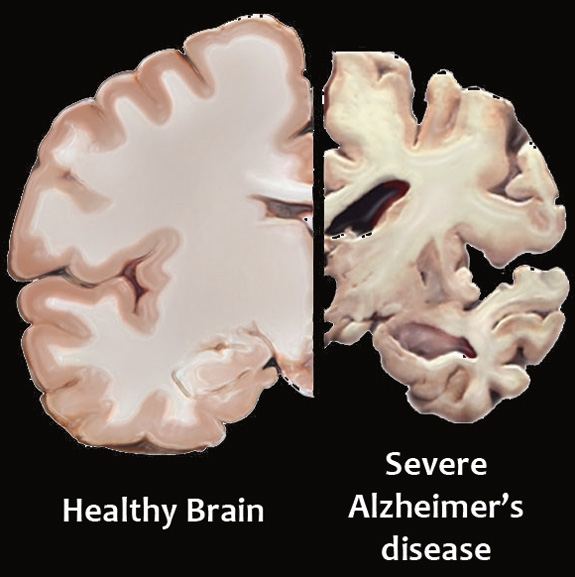
Because of gene insertions, this mouse model makes slightly more amyloid-beta than normal animals and the protein is over-expressed only in the neurons rather than throughout the body. This begins during the third week of development. The mice end up with a series of deficits that are just like human Alzheimer’s. “The first injury in Alzheimer’s is the loss of synaptic connections,” Dr. Galvan says. “That is something our mice recapitulate.”
Drs. Galvan and Oddo are also collaborating with the laboratory of Rochelle Buffenstein, Ph.D., and in particular her Ph.D. student, Yael Edrey, to look at the aging brain of the longest-lived rodent, the East African naked mole rat. “As part of Yael’s Ph.D. work, she is examining amyloid-beta plaques and tau-protein tangles in both mice and mole rats of different ages,” says Dr. Buffenstein, professor of physiology who moved her research colony of naked mole rats to the Barshop Institute in 2007. “It is often speculated that mice do not live long enough to naturally acquire Alzheimer’s disease. The naked mole rat may be a natural model for this debilitating disease.”
Meanwhile, the transgenic mouse models used in the Oddo and Galvan research studies are strikingly representative of human Alzheimer’s progression. Because of the information they are providing, these rodents may be the best hope for stopping the memory-robbing disease in the future.
