 What do frequent stoppages in breathing during sleep do to the body? A team of 30 researchers, led by three senior investigators in the Graduate School of Biomedical Sciences, is seeking answers in a unique model of sleep apnea in rodents. A five-year, $9.5 million grant from the National Heart, Lung, and Blood Institute of the National Institutes of Health (NIH) supports the research program in the departments of Pharmacology, Physiology and Anesthesiology. The multidisciplinary team is mimicking sleep apnea in the laboratory by simulating the sporadic reductions in oxygen that mark the disorder. In special chambers, the rodents breathe a normal oxygen level, then half of that level, in alternating cycles for eight hours — the course of a normal night’s sleep.
What do frequent stoppages in breathing during sleep do to the body? A team of 30 researchers, led by three senior investigators in the Graduate School of Biomedical Sciences, is seeking answers in a unique model of sleep apnea in rodents. A five-year, $9.5 million grant from the National Heart, Lung, and Blood Institute of the National Institutes of Health (NIH) supports the research program in the departments of Pharmacology, Physiology and Anesthesiology. The multidisciplinary team is mimicking sleep apnea in the laboratory by simulating the sporadic reductions in oxygen that mark the disorder. In special chambers, the rodents breathe a normal oxygen level, then half of that level, in alternating cycles for eight hours — the course of a normal night’s sleep.
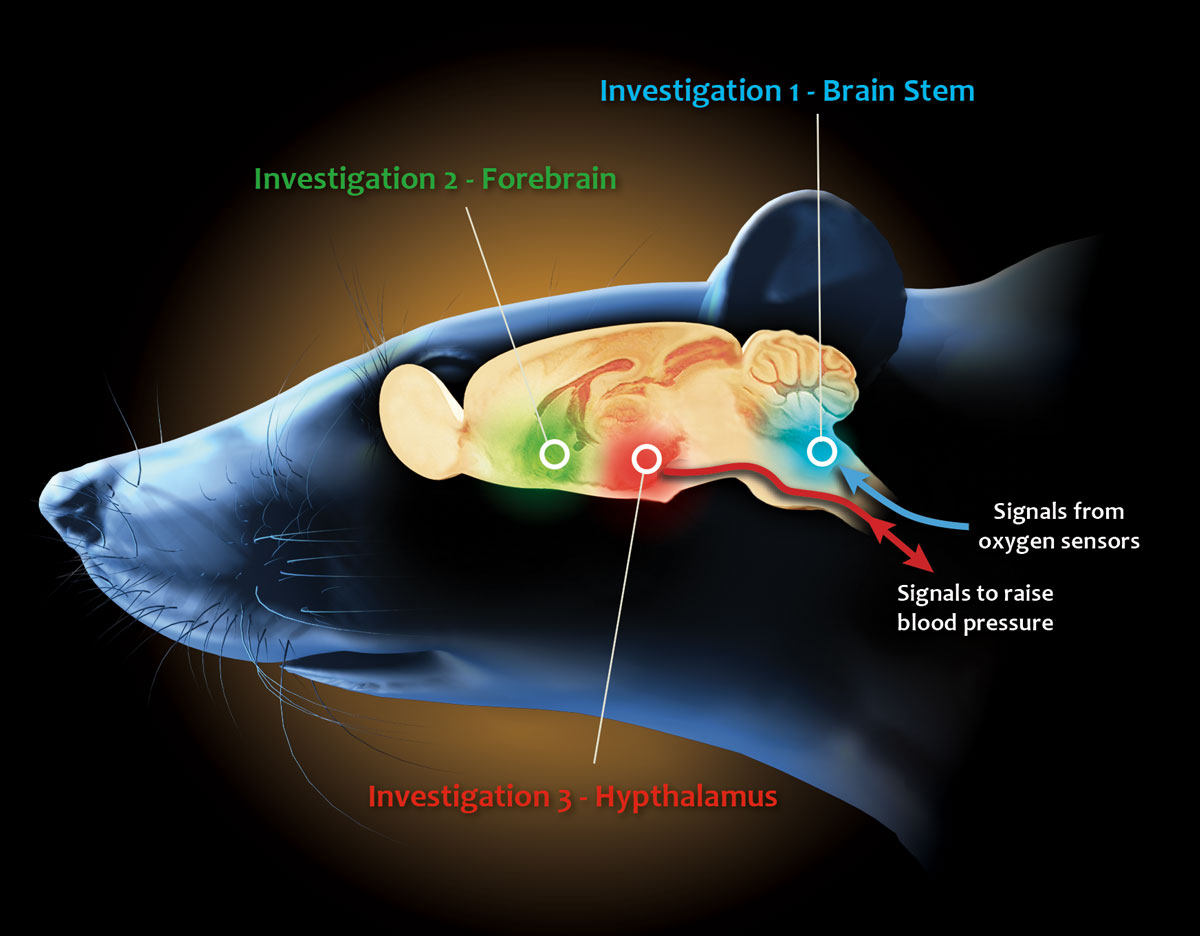
First team observes brain stem
The Health Science Center researchers, initially supported by seed money from the university’s Presidential Research Enhancement Fund leading to the NIH grant, are divided into three teams categorizing different changes that take place in the brain and nervous system with recurring low oxygen. The grant principal investigator, Steven W. Mifflin, Ph.D., professor of pharmacology, leads a contingent studying what happens when oxygen sensors in the arteries inform the brain that blood oxygen levels have fallen.
“We are looking at how that information is processed in the brain stem and how the processing is altered after repetitive exposure to low oxygen levels in blood,” Dr. Mifflin said. “You have these oxygen sensors telling the brain that levels have fallen, resulting in faster breathing, raised blood pressure and other actions to try to get more oxygen into your blood. Part of the reason sleep apnea patients have high blood pressure is the repetitive activation of this machinery.”

Sleep apnea simulator
The chambers, designed by the Division of Instrumentation Services at the university, rely on a series of timers that control the flow of gasses through valves, allowing nitrogen to enter and displace some of the oxygen while the rodents sleep. The rodents experience about 80 episodes of reduced oxygen over an eight-hour sleep cycle, which is during the daytime because the rodents are nocturnal. “The model we are using is a fairly moderate form of sleep apnea,” Steven W. Mifflin, Ph.D., said. Most people have this form, he said, but there are severe cases in which an individual may stop breathing every couple of minutes, or 30 times in an hour.
Second team observes forebrain
Tom Cunningham, Ph.D., associate professor of pharmacology, leads a second team investigating how forebrain mechanisms contribute to elevated blood pressure in the rodents, even when the blood oxygen levels have returned to normal. “The reason blood pressure remains high, even after oxygen levels return to normal, is because the brain adapts,” he said. “When oxygen is low, genes induce changes in brain function, which stimulate the heart and arteries to maintain high blood pressure. We are observing these changes in the rodents experiencing intermittent low oxygen conditions.”
Blood pressure increases by the same amount in these animals as it does in people with sleep apnea, he noted. “Dr. Mifflin developed this rodent model, which has a great deal of potential clinical relevance,” Dr. Cunningham said. “I think that is why the Health Science Center received this large and important NIH grant.”
Third team evaluates hypothalamus
A third investigator, Glenn Toney, Ph.D., associate professor of physiology, leads a team evaluating how an area of the hypothalamus processes input from both oxygen sensors in the arteries and the genes triggered by low oxygen. The hypothalamus governs the sympathetic nervous system, which causes blood vessels to constrict and increases the pumping force of the heart, further contributing to the high blood pressure.
“It turns out information from arterial oxygen sensors transmit signals to the brain stem, which communicates with the hypothalamus,” Dr. Toney said. “Meanwhile, the forebrain neurons Dr. Cunningham is studying also communicate with the hypothalamus. According to our hypothesis, the hypothalamus integrates information from both the brain stem and the forebrain, thereby activating brain stem sympathetic nerve activity, which results in high blood pressure.”
This NIH-funded research team is supported by several institutional core facilities. Carmen Hinojosa-Laborde, Ph.D., adjunct associate professor of anesthesiology, leads the animal core, which is responsible for exposing rodents to the sleep apnea model. David A. Morilak, Ph.D., professor of pharmacology, leads the biochemical and molecular core, which ensures standardization of the tests that investigators run to verify findings. Dr. Cunningham leads the neuroanatomy core, which performs anatomical analyses using standardized procedures.
Health Science Center support results in NIH grant
The Health Science Center has been extremely supportive of these cores, the construction of 10 chambers and the recruitment of faculty for the project, Dr. Mifflin said. “The seed money we received from the university’s Presidential Research Enhancement Fund totaled $125,000. That initial investment has resulted in a $9.5 million grant.”
Study has enormous implications
The true result is an unprecedented research study of one of our nation’s most prevalent – and debilitating – disorders of sleep. The study is a multipronged effort to unravel the mystery of the “black box” of sleep apnea, the basic biology of this disease. The implications are enormous. “Sleep apnea is rampant, absolutely huge, particularly here in South Texas, because obesity and diabetes are both associated with sleep apnea, and we have a preponderance of both conditions,” Dr. Toney said. “In addition, one in three people in the U.S. is hypertensive. Sleep apnea is a complex issue, and this research is being conducted with considerable technical sophistication. People with real expertise are working here on it.”
