They’re in every cell in every living organism, and now researchers have a better understanding about heat shock proteins, also called chaperone proteins, and their role in human diseases.
The proteins were first identified in cells subjected to heat. They help the body maintain proper protein function and, importantly, prevent the accumulation of damaged proteins. This accumulation is thought to be important in the development of diseases of the brain such as Alzheimer’s and Parkinson’s.
Heat shock proteins with a molecular weight of 70 can break apart protein complexes rather than simply binding to them. Called Hsp70s, they collide with them and generate a force that dissolves the complexes.
“No one knew how the heat shock proteins pull apart bad protein complexes,” said Rui Sousa, Ph.D., a professor of biochemistry in the School of Medicine who authored a study with biochemistry professor Eileen M. Lafer, Ph.D., in the journal Nature Structural & Molecular Biology.
“At the molecular level, everything is moving, colliding and bumping, and smashing into other components of the cells. We found that the system moves Hsp70s to where they are needed. Once this occurs, collision pressures pull things apart.”
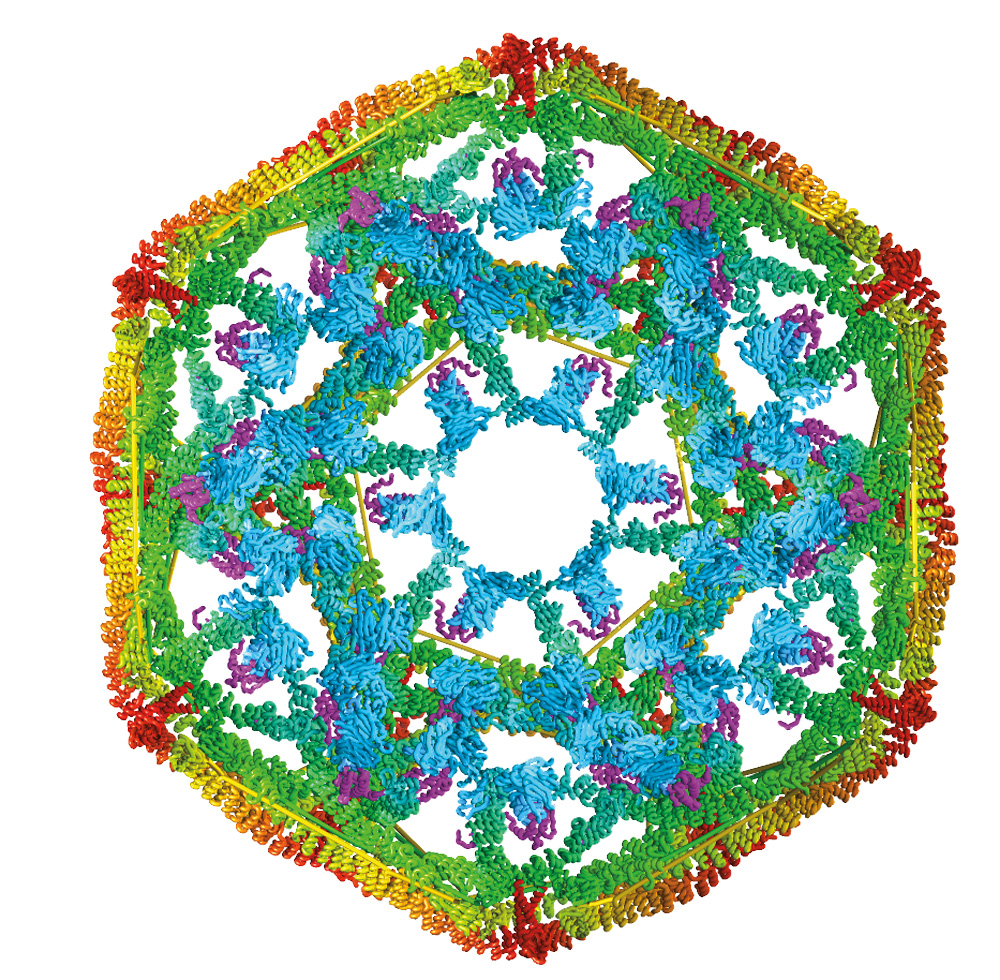
They studied clathrin, a protein that is uniform in size and shape and is important in making intracellular cages that transport other proteins.
Dr. Lafer grew clathrin “cages”—shaped like microscopic soccer balls—that provided the biological raw material for Dr. Sousa and the team to study the force that occurs with Hsp70 collisions. The clathrin model system could be manipulated to yield precise results.
By making variants of clathrin with recombinant DNA technology, team members were able to manipulate this biological material in ways that allowed them to determine the mechanism by which it is taken apart by Hsp70.
“This work was a tour de force, requiring the convergence of exceptional biochemical and molecular genetic skills with a deep understanding of the principles of physical chemistry,” said Bruce Nicholson, Ph.D., chair of the Department of Biochemistry. “Such insights into the most basic aspects of protein chemistry and cell biology are often, as in this case, driven by a curiosity to find out how the molecular machines that drive our bodies work. But from these basic pursuits of scientific curiosity will often stem great benefits to human health.”
Understanding Hsp70 behavior may have relevance to human disease. By increasing Hsp70 function, scientists cured Huntington’s, a neurodegenerative disease, in a fly model. Tumors rely on Hsp70s to survive, so lowering Hsp70 function also is a topic in cancer research.
